 I can’t remember just at what point in my studies around 1998-2005 I discovered what a truly amazing thing WATER is.
I can’t remember just at what point in my studies around 1998-2005 I discovered what a truly amazing thing WATER is.
And we just take it for granted.
Water is unlike nearly every other substance. Consider:
- It can dissolve or dilute nearly everything, and one way or another you can get water back again.
- It exists naturally in solid, liquid and gaseous states
- It forms up to 78% of our bodies
- It covers 71% of Earth’s surface
- It has a cycle that keeps the amount of water on Earth more or less constant – clouds, rain, streams, river, sea, clouds… with little extras like groundwater and ‘drinking’ for most living things on the planet too.
- Its surface tension enables it to act as a solvent – lifting off dirt from clothes, for example.
- Its high heat capacity makes it immensely important as a regulator of Earth’s temperature – keeping most of it in a range ‘comfortable’ for life.
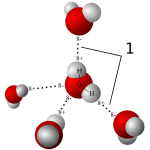 As you know, water is made from two Hydrogen atoms and one Oxygen, hence its chemical formula H2O (that two should be dropped below the line, but that’s the best I could do).
As you know, water is made from two Hydrogen atoms and one Oxygen, hence its chemical formula H2O (that two should be dropped below the line, but that’s the best I could do).
The bond between the hydrogen and oxygen atoms leaves it with a slightly positive charge, which is what helps it bond weakly with other substances and with other water molecules. Adding e.g. washing powder, or increasing the electrostatic charge through using wash balls, changes the surface tension and makes its washing capacity more efficient. It took me ages to understand why these wash ball gizmos work. They also help reduce the deposition of limescale in your washing machine, if you’re in a hard water area.
 Water is not easily compressed, which is why it is brilliant for use in pumps and for hydraulic engineering.
Water is not easily compressed, which is why it is brilliant for use in pumps and for hydraulic engineering.
And water, one way or another, features in nearly all of my A to Z posts this year. Why not have a glass of it now?! Cheers!
More amazing stuff on water at wikipedia.
Pictures: hydrogen bonds with water from wikipedia; interference waves on the Gironde coast (mine); mist, sea, ice and mountains at Svalbard (mine)


Fascinating. Water is one of those things you take for granted but is, obviously, absolutely vital.
We couldn’t live without it!
Cheers! It’s lunchtime, so off for my glass of water right now.
Cheers!
Its the finest drink and helps make some good one too! Cheers
Indeed – supping one right now 🙂
I’ve lived my whole life surrounded by water. It’s an odd concept to me to go somewhere that doesn’t have giant lakes around the state.
~Patricia Lynne aka Patricia Josephine~
Member of C. Lee’s Muffin Commando Squad
Story Dam
Patricia Lynne, Indie Author
Yeah, I live on an island, which gives one a certain affection for the stuff. 😀
Thanks for this nice post! It’s what makes Earth the blue planet – so beautiful from space – and supports all of life!
Indeed. Remember when we first saw that amazing view of earthrise from the moon? I imagine kids today take it for granted…
You’re right. Water is a common, but amazing substance.
Thanks, Sonia!
The idea of taking water for granted reminds me of our trip to New Zealand years ago. Hiking in the rather damp Fjiordland area, we met a young Israeli hiker who couldn’t stop raving about all that water…just being allowed to flow out to sea and go to waste! If you live in a desert, you don’t take water for granted. Most of California hasn’t twigged to the fact that most of California is a desert.
Yes. I’ve been to Mono Lake.
Water has no colour, smell or taste and yet it has amazing properties. I keep coming back to it as something to blog about.
Sue’s Trifles
Hi Jemima – it is an amazing substance isn’t it .. and thankfully I drink a lot of it – so am pleased it’s around. I think I learnt a bit about the wash ball … and will pay more attention when I see one around … cheers for now – Hilary
PS – sorry just way behind with the A-Z returning comments and now too finding your wonderful subject …